10 Tips on How to Prepare for a GMP Inspection
10 Tips on How to Prepare for a GMP Inspection
Introduction
GMP inspections are a cornerstone of pharmaceutical regulation, ensuring that manufacturers consistently meet the required standards of quality, safety, and efficacy. The primary objective of these inspections is to protect public health by preventing contamination, manufacturing errors, and systemic failures that could compromise patient safety.
Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) conduct GMP inspections to verify compliance with applicable laws and guidelines. These inspections typically cover facilities, equipment, documentation, personnel training, and adherence to Standard Operating Procedures (SOPs).
This topic outlines ten practical, field-tested steps pharmaceutical companies can take to prepare effectively for a GMP inspection—whether announced or unannounced—while fostering a sustainable culture of compliance and continuous improvement.
1. Conduct Thorough Internal Audits
Internal audits form the foundation of GMP inspection readiness. They allow organizations to proactively identify gaps, deviations, or weaknesses before regulators do.
By conducting structured self-assessments, companies can implement Corrective and Preventive Actions (CAPA) early, reducing inspection risk and reinforcing compliance discipline across the organization.
Key Internal Audit Focus Areas
- Facilities
Verify cleanliness, HVAC performance, and facility design controls that prevent cross-contamination.
Example: HEPA filter integrity and particle count compliance in sterile areas. - Equipment
Review calibration, qualification, and maintenance records.
Example: An uncalibrated tablet press may cause dosage variability—a critical GMP violation. - Documentation & Records
Ensure batch records, deviations, and change controls are complete and contemporaneous. - Personnel Training
Confirm training effectiveness, not just completion. Inspectors may question any operator on GMP fundamentals.
Actionable Steps
- Schedule audits quarterly or semi-annually
- Use cross-functional audit teams
- Audit against regulations, prior findings, and industry best practices
2. Ensure All Documentation Is Up-To-Date and Accessible
Documentation is the tangible proof that GMP systems are under control. Inadequate or outdated documentation is one of the most common causes of inspection observations.
Critical Documents to Control
- Batch Manufacturing Records
- SOPs aligned with real practices
- Equipment logbooks
- Deviations, investigations, and CAPA records
Actionable Steps
- Periodically review and approve SOPs
- Implement electronic document management systems (EDMS)
- Train staff on real-time documentation discipline
3. Maintain Cleanliness and Organization
Cleanliness is not cosmetic—it is a regulatory requirement directly tied to product safety.
Areas of Emphasis
- Production and cleanroom areas
- Warehousing and material segregation
- Equipment cleaning and status labeling
Actionable Steps
- Define daily, weekly, and periodic cleaning schedules
- Document all cleaning activities
- Use visual controls and labeling systems
4. Form a Dedicated Inspection Team
A structured inspection team ensures clarity, speed, and confidence during regulatory interactions.
Key Roles
- Inspection Point of Contact
- Subject Matter Experts (SMEs)
- Document Controllers
Actionable Steps
- Assign roles well in advance
- Train the team using mock inspections
- Ensure rapid document retrieval capability
5. Analyze Reports From Previous Inspections
Historical inspection data is one of the most valuable preparation tools.
Focus Areas
- Recurring deficiencies
- CAPA effectiveness and sustainability
Actionable Steps
- Track previous findings
- Verify inspection commitments were fulfilled
- Prepare evidence of improvement
6. Train Staff on Inspection Protocols
Inspectors often evaluate culture through employee behavior and knowledge.
Training Focus
- GMP fundamentals by role
- How to answer inspection questions
- Professional conduct and escalation paths
Actionable Steps
- Conduct mock interviews
- Use role-playing scenarios
- Reinforce “answer what you know—nothing more”
7. Create a Detailed Inspection Readiness Plan
Inspection readiness is a continuous system, not a last-minute effort.
Core Plan Components
- Defined tasks and owners
- Timelines and deliverables
- Periodic readiness reviews
Actionable Steps
- Develop a master GMP readiness checklist
- Integrate readiness into the QMS
- Review status in management meetings
8. Conduct Mock Inspections and Use External Expertise
Mock inspections simulate regulatory pressure and expose blind spots.
Benefits
- Objective gap identification
- Staff confidence building
- Reduced inspection anxiety
Actionable Steps
- Engage experienced GMP consultants
- Perform routine internal mock audits
- Implement corrective plans immediately
9. Plan a Logical Inspection Route
Inspection flow influences efficiency and perception.
Why It Matters
- Minimizes operational disruption
- Ensures access to critical areas
- Demonstrates organizational control
Actionable Steps
- Map inspection routes in advance
- Align document access points
- Perform trial walkthroughs
10. Set Up a War Room
Essential Elements
- Organized documentation access
- Communication tools
- Availability of key personnel
Actionable Steps
- Prepare the room before inspection day
- Categorize documents logically
- Ensure SMEs are on standby throughout
Frequently Asked Questions (FAQ)
What documents should be readily available during a GMP inspection?
Batch records, SOPs, training records, deviation reports, CAPA documentation, and equipment logs.
How should deviations and CAPAs be handled before inspection?
All deviations must be investigated, closed, and supported with effectiveness checks.
How can companies stay ready for unannounced inspections?
By maintaining continuous compliance, regular audits, and active training programs.
What role does senior management play?
Leadership sets compliance tone, allocates resources, and demonstrates accountability.
Conclusion
Preparing for a GMP inspection is a structured, organization-wide effort that goes far beyond inspection day. Internal audits, strong documentation systems, trained staff, mock inspections, and leadership engagement collectively ensure sustained compliance.
The real objective is not simply passing inspections—but embedding GMP principles into everyday operations. Organizations that achieve this earn regulator trust, protect patients, and strengthen long-term business resilience.





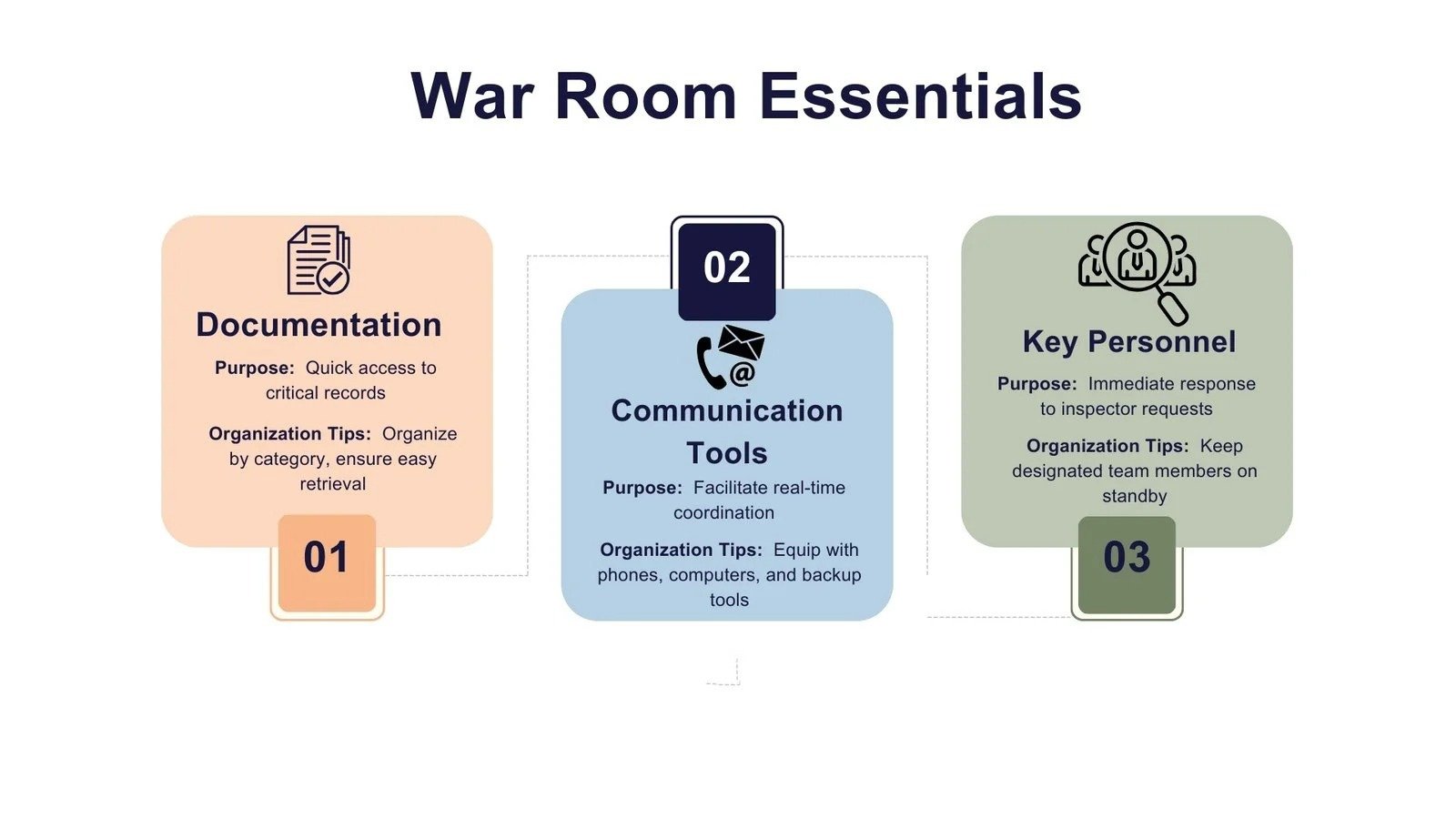 A war room is the operational command center during an inspection.
A war room is the operational command center during an inspection.